Rocks, Minerals and Gems
Most metals do not exist in nature as the metal. While a few can be found in their metallic form, such as gold, silver, and the other noble metals, most metals are found as compounds. This is because the metals are easily oxidized to give metal (M) cations, Mn+, which do not exist independently. They must be combined with other species that are negatively charged. The positive charge on the metal ion must be compensated by a negative charge on some anion in order to produce compounds that overall have no net charge. Some of the most common ions that combine with metals are anions of single atoms such as the halides (F-, Cl-, Br- and I-), oxide (O2-) and sulfide (S2-) but other ions comprised of several atoms are also common. These include carbonate (CO32-), nitrate (NO3-), sulfate (SO42-), silicate (SiO44-) and phosphate (PO43-). Particularly common are the oxides of aluminum (alumina or Al2O3) and silicon (silica or SiO2), as well as minerals that contain hybrids of these known as aluminosilicates.
These metal compounds form the basis of a number minerals that important for a variety of applications. They include the precious and semi-precious gemstones such as rubies, emeralds, sapphires, quartz, citrine, amethyst and beryls, but they also encompass building stones such as granite, sandstone, limestone, marble and alabaster - materials that were also used for statuary and other decorative objects. What distinguishes rocks and stone from gems is that the latter are single crystals with very precise arrangements of the atoms or ions in the crystal lattice. This makes gems transparent as long as they are made of very good single crystals. The presence of defects in the crystals will be noticeable, and a sample will not be as clear and transparent if it contains many defects. This is a result of the way light reflects off the interfaces where crystal defects occur.
Stone, on the other hand, is largely made of aggregates of very small crystals, often called grains. These may be composed of a variety of different mineral substances compacted together, and stones are usually opaque. This is the case for granite and sandstone. Granite is an igneous rock formed by volcanic activity composed primarily of quartz and feldspar that have been heated together. Sandstone is a sedimentary rock also composed mostly of quartz and feldspar with grains that are less than 2 mm is size and that have been compacted under high pressure. While granite and sandstone are known more for their architectural usage, they have also been employed in sculpture.




The Carbonate Minerals
Limestone, marble and alabaster are all based on colorless calcium carbonate, CaCO3 (also known as calcite), or its close relative dolomite, a carbonate mineral containing both calcium and magnesium ions. The fundamental chemistry of carbonate compounds is very important here and helps us understand the degradation of sculpture and architecture made of limestone and marble due to acid rain.
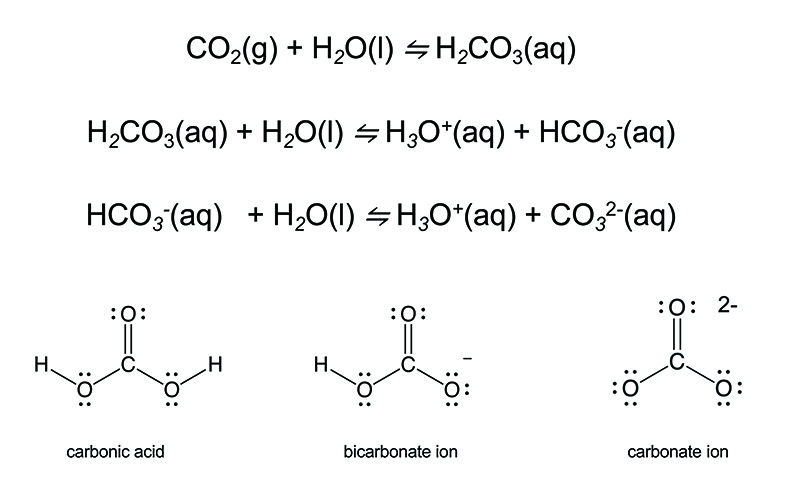
The carbonate ion arises when carbon dioxide, CO2, the substance that is the main oxidation product of all organic matter and the gaseous byproduct of our bodies' energy consumption of plants and animals, dissolves in water. CO2 dissolves reversibly in water to produce carbonic acid, H2CO3, which is also the substance responsible for the fizz of carbonated beverages. Such beverages are under slight pressure, which helps to keep the CO2 dissolved but the carbon dioxide will escape the water and the drinks will go flat. But there is another equally important reaction of carbonic acid, and that is it ability for the hydrogen atoms to dissociate from the molecule as protons, H+, in water. Often these are written as hydronium ions, H3O+, because the H+ ions will readily bond to the electrons on the oxygen atom of water. And this process can happen twice. In the first case, the result is the bicarbonate ion, HCO3-, and the second dissociation gives CO32-. These steps are promoted by the addition of base, but they are also reversible upon addition of acid.

When calcium is oxidized, it forms calcium oxide, CaO, which is basic and reacts with water to form calcium hydroxide, Ca(OH)2. This compound dissolves in water to produce Ca2+ and OH- ions. If carbon dioxide is present, it will react with the hydroxide ions to form first the bicarbonate ion, and if the water is sufficiently basic, it will react further to form the carbonate ion. In Nature, calcium carbonate will ultimately precipitate and form chalk or limestone. Under sufficient pressure, the limestone will be compressed to form marble and alabaster. The White Cliffs of Dover are famous examples of an impressive formation of chalk along the southern coast of England. The mineral name of CaCO3 is calcite. It adopts a rhombohedral lattice. A mineral with the same structure but having magnesium ions (Mg2+) replacing some of the Ca2+ is known as dolomite.
Once carbonate minerals such as limestone, marble and alabaster, they will be exposed to the chemical compounds present in the air. Rain is naturally acidic because of the presence of carbon dioxide that dissolves in the suspended droplets of water, but it has become more acidic since the Industrial Revolution owing to the production of large quantities of sulfur and nitrogen oxides as well. This lower the pH of rain substantially, and the acidic rain water will erode any exposed carbonate mineral surfaces.
Gemstones
Gems are crystalline materials in which the atoms or ions arrange themselves in regular arrays in a highly crystalline lattice. They are valued for their clarity as well as their ability to reflect light when properly cut. There are several important classes of gemstones presented here. Several of these are based on their chemical composition as well as crystal structure. Diamonds are made of carbon and are a unique class, while there are three general classes based on other chemical substances: silica (SiO2), alumina (Al2O3) and aluminosilicates, which are more complex structures involving aluminum and silicon oxo-anions. The silica-based minerals include quartz, citrine and amethyst, while the alumina-based gems are ruby and sapphire. Alumino-silicate minerals incorporate berylium include the beryls, which encompasses emeralds and aquamarine as well as some other less-well known gems. Jade is interesting in that there are two different minerals that are referred to as jade - nephrite, which is a silicate of calcium and magnesium, and jadeite, which is sodium aluminosilicate. Turquoise is a blue or blue-green copper phosphate mineral that differs from these other classes in not forming transparent crystals. Rather, it is an opaque mineral that is also known for the inclusion of other mineral types, giving rise todark regions embedded in the blue backgrounds. Pearls are unique in being produced biologically from several classes of mollusks. More information including the compositions and structures of these gemstones are found in the links below.
Most gemstones are recognized for their use in jewelry and other costly decorative art objects such as crowns, jewel-encrusted sword hilts, religious objects such as chalices and crosses, among others. The classification of whether something is a precious or semiprecious gemstone depends largely on its cost, which can vary from period to period. In recent times, the availability of synthetic versions of gems has affected their market value. In some cases, a completely different crystalline material has served as a substitute for something much more expensive. This is true in the case of cubic zirconia - ZrO2 - which is used widely as a substitute for diamonds at a small fraction of the cost.
One other naturally-occuring carbonate material prized as a gemstone is the pearl. Unlike the other minerals that are produced as in the earth's crust, pearls are made by mollusks in a biodeposition process in which small crystallites of CaCO3, mostly as the mineral form aragonite with small amounts of calcite, are deposited in a matrix of chitin, lustrin and various silk-like proteins.
Learn More
Shortcuts
Credits for the Header Image
- Artist Unknown, Possibly Indian or Spanish, imported France, Parure, 1780–1820, France, Medium Emeralds, diamonds and yellow gold with silver overlay, from the collection of the MFAH.
- Unknown Central and South American, Pair of Tubular Inlaid Ear Ornaments, 100–700 AD, Gold and turquoise, from the collection of the MFAH.
- unknown Indian, Gem-Set Bottle, Mid-17th century, Gold with diamonds, emeralds and rubies, from the collection of the MFAH.
- Unknown Central and South American, Axe-God Celt, 500–1000 AD, Jade, from the collection of the MFAH.
Credits for the Minerals links
- Diamond: The Hope Diamond, National Natural History Museum, Smithsonian Institution.
- SiO2-based Gemstones: Quartz, Photo by Pet Kratochvil, Public Domain.
- Al2O3-based Gemstones: A collection of colored spinels. Photo: Robert Weldon/GIA, Dr. Eduard J. Gübelin Collection, Gemological Institute of America (GIA).
- Alumino-silicate-based Gemstones: Red beryl from the Wah Wah Mountains, Beaver County, Utah. Photo by Parent Géry, Wikimedia Commons, Public Domain.
- Jade: Unknown Chinese, Box and Cover, 18th–19th century, Jade from the collection of the MFAH, Public Domain.
- Turquoise: Unknown North American, Bracelet, 1900–1945, Silver, turquoise.
- Pearls: Pendant, 19th century, Pearl, gold, and enamel, from the collection of the MFAH, Public Domain.
- Cutting Gemstones: Gemstone Cuts, Public Domain.
Department of Chemistry

Houston, TX
6100 Main St., Houston, TX 77005-1827 | Mailing Address: P.O. Box 1892, Houston, TX 77251-1892 713-348-0000 |